17 ways to produce hydrogen sustainably

Hi everyone – I’m sorry for falling off the map after that last post. I have been a bit busy taking care of some things, including physically moving to Denver. The move was a little unplanned but thoroughly welcomed. One of my goals this year was to explore new networks and Denver will be a great hub to do that from. I’ve been incredibly impressed with the Denver tech community + the many exciting things happening around energy transition and climatetech in this city. The easy flights to Houston are also a big plus!
You might have also seen that I was made an advisor to Boundless Capital Partners, a Denver-based investment bank focused on energy infrastructure and technology. I’ll continue to “do my own thing” but this gives me chance to support a stellar team taking a differentiated approach to investment banking while also staying adjacent to some relevant dealflow. Give me a shout if you’d like to learn more or to generally catch up.
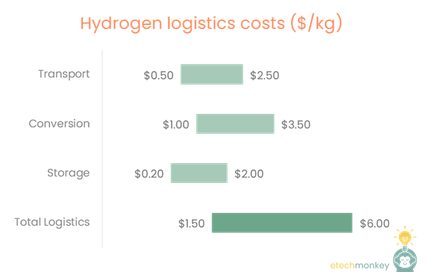
Today let’s talk hydrogen production.
A lot of you might know that the reason why I am long hydrogen, despite the logistical shortcomings of the current network, is the many ways that hydrogen is able to be produced from a variety of sources. Hydrogen is the simplest molecule in the universe, which lends itself to be a product or by-product of a variety of different pathways.
See below for some of the different ways I know of to produce low-emissions hydrogen (Note that these are not vetted or sorted. This is just a list I compiled of advertised hydrogen production methods from various companies, mostly startups.):
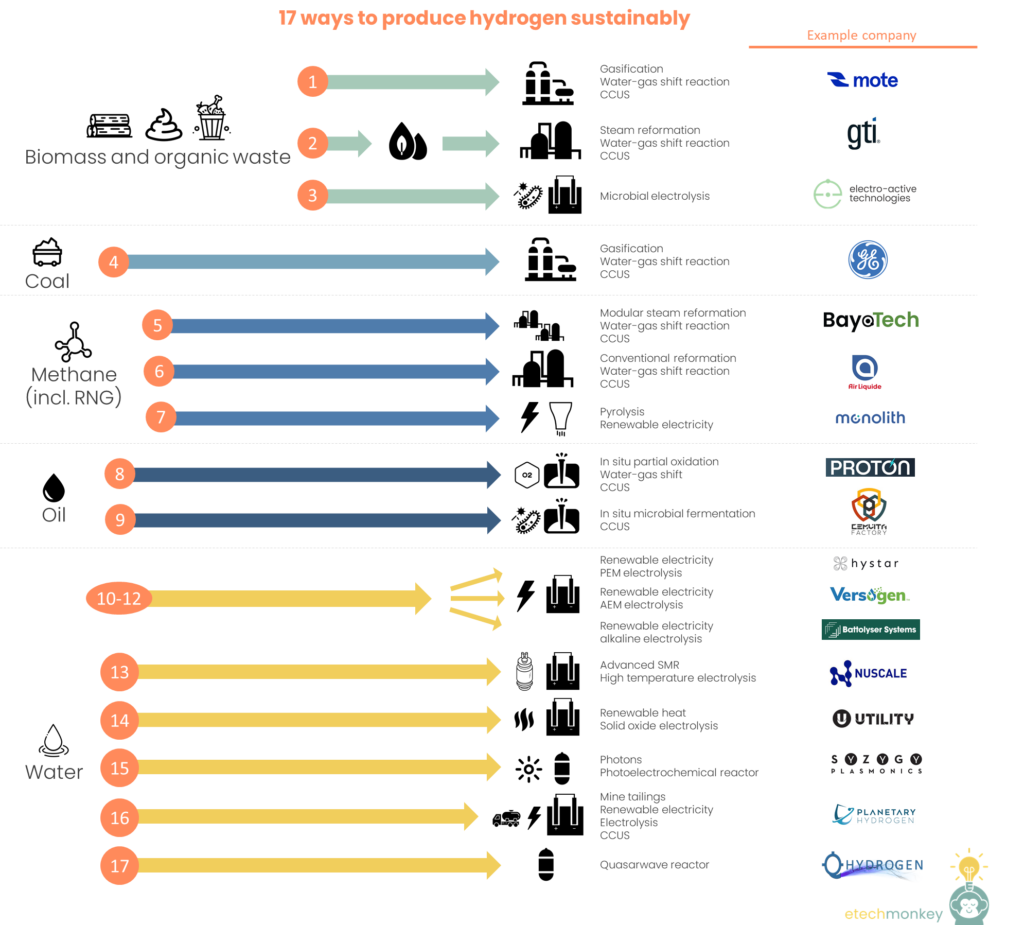
- Biomass gasification + heat + water-gas shift + CCUS | Mote
- Biomass -> biofuel steam reforming + heat + water-gas shift + CCUS | GTI
- Biomass / waste + renewable electricity + microbial electrolyzer | Electro-Active Technologies
- Coal gasification + heat + water-gas shift + CCUS | GE
- Modular steam methane reformation + heat + water-gas shift + CCUS and/or RNG | Bayotech
- Conventional SMR, partial oxidation or autothermal reforming of methane + heat + water-gas shift + CCUS and/or RNG | Air Liquide
- Methane pyrolysis + renewable electricity + possibly RNG | Monolith Materials
- In situ partial oxidation of hydrocarbons + water-gas shift + CCUS | Proton Energy
- In situ oil + fermentation by microbes + CCUS | Cemvita
- Water + renewable electricity + PEM electrolyzer | Hystar
- Water + renewable electricity + AEM electrolyzer | Versogen
- Water + renewable electricity + alkaline electrolyzer | Battolyser
- Water + advanced SMR + high temperature electrolyzer | NuScale
- Water + renewable heat + solid oxide electrolyzer | Utility Global
- Water + photons + photoelectrochemical reactor | Syzygy
- Water + renewable electricity + mine tailings + electrolyzer + CCUS | Planetary Hydrogen
- Water + quasar wave reactor | Q Hydrogen
*Note: The list could be made even more precise through differentiating between SMR, electrolysis, and pyrolysis by type of catalyst used. For example, there is a sub-category of plasma-based catalysts that are used in pyrolysis.
For a more in-depth explanation of some of these, the DOE is a great resource.
A few observations on the list:
- Most new methods rely on water as a primary feedstock. Not many companies that I could find are working on new implementation of gasification or reformation from hydrocarbon sources.
- Electrolysis as a broad category is filled with players ranging from large public companies to small startups. The larger companies are focused more on the more mature technologies, which, for electrolysis is alkaline and, to a lesser extent, PEM. Startups tend to be working more on a mix of PEM, AEM, and solid oxide.
- There is a lot of competition in electrolysis technology. Electrolytic methods tend to be evaluated based on:
- Cost and availability of secondary inputs (heat or electricity or both, catalysts, membrane, electrolyte)
- Ability to output pressurized hydrogen
- Current density (which, to my understanding, is proportional to power density and thus hydrogen generation rate, so generally speaking, higher current density = larger hydrogen generation in the same amount of area)
- Partial load range (which is wider for PEM and solid oxide than for alkaline and AEM)
- Long term stability and durability of the system (alkaline tends to be better tested in this arena)
- Ability to scale (modularity and absolute size able to be achieved)
- Overall efficiency (electrolysis usually lands at ~60-80%)
It’s my understanding that no one electrolytic method wins in all or even the majority of these variables, which makes “choosing a winner” in this arena a complex optimization problem. With more complexity in choice comes more competition for the same business. The higher competition is somewhat compensated by the larger number of players interested in electrolytic production of hydrogen vs. other methods.
- An emerging category of hydrogen production is biohydrogen – or the production of hydrogen via biological methods. This can range from producing hydrogen with algae or using a microbial-based reactor (like microbial electrolysis). I had a difficult time finding startups that were working on commercializing this technology. It seems that most of the development in this area is being done by universities. With the amount of capital going into microbiology, and especially microbiology in energy transition (currently focused on developing synthetic oils and chemicals), we should see biotech startups emerging focused on producing hydrogen as well.
Hope you enjoyed today’s roundup. Would love any feedback or comments!